
Ubiquitin & autophagy: safeguarding cellular homeostasis
The Ubiquitin System
The ubiquitin system is a versatile signaling pathway in which the small protein ubiquitin is covalently attached to target proteins. This process involves an enzymatic cascade (E1 activating enzymes, E2 conjugating enzymes, and E3 ligases) and creates a molecular “tag” that can alter a protein’s fate: marking it for proteasomal degradation, changing its activity, or guiding it to specific cellular pathways. Deubiquitylases (DUBs) terminate ubiquitin signals (and we love them).
Autophagy
Autophagy is a lysosome-dependent degradation pathway that recycles cellular components. In selective autophagy, ubiquitin acts as a key signal: damaged or superfluous organelles and protein aggregates are tagged with ubiquitin, recognized by autophagy receptors, and engulfed by autophagosomes, which then fuse with lysosomes for degradation. Together, the ubiquitin system and autophagy form a central quality-control network, ensuring that proteins and organelles are properly maintained, recycled, or removed.
When these pathways fail, toxic proteins accumulate and dysfunctional organelles persist, driving the development of human disease. Defects in ubiquitin signaling and selective autophagy are linked to neurodegeneration (e.g. Parkinson’s, ALS), cancer, metabolic disorders, and infections, highlighting their central role in health and disease.
Our research projects
-
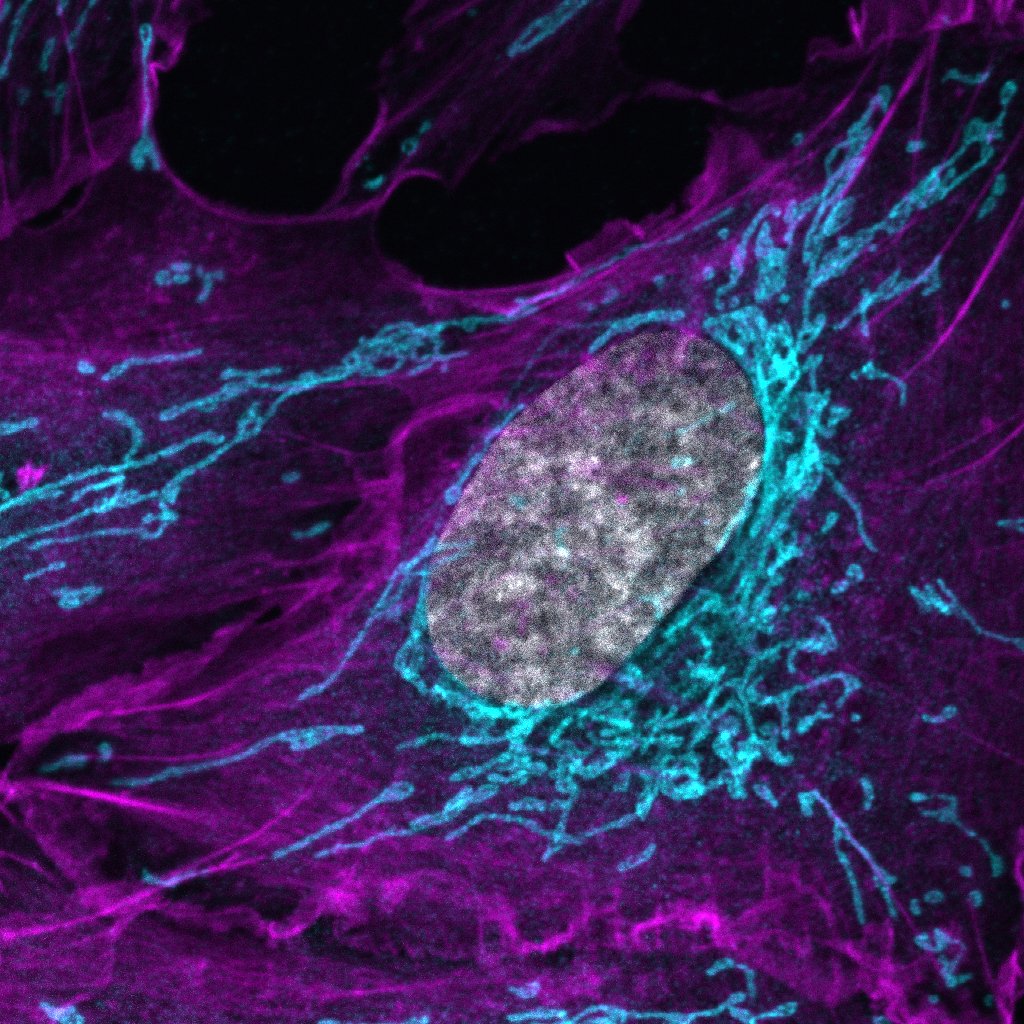
Ubiquitin signaling in selective autophagy
Our group investigates ubiquitin-dependent mechanisms that govern selective autophagy. We aim to understand how ubiquitin signals, driven by E3 ligases and counteracted by DUBs, coordinate cargo recognition and organelle clearance, and how disruption of these pathways contributes to disease.
-
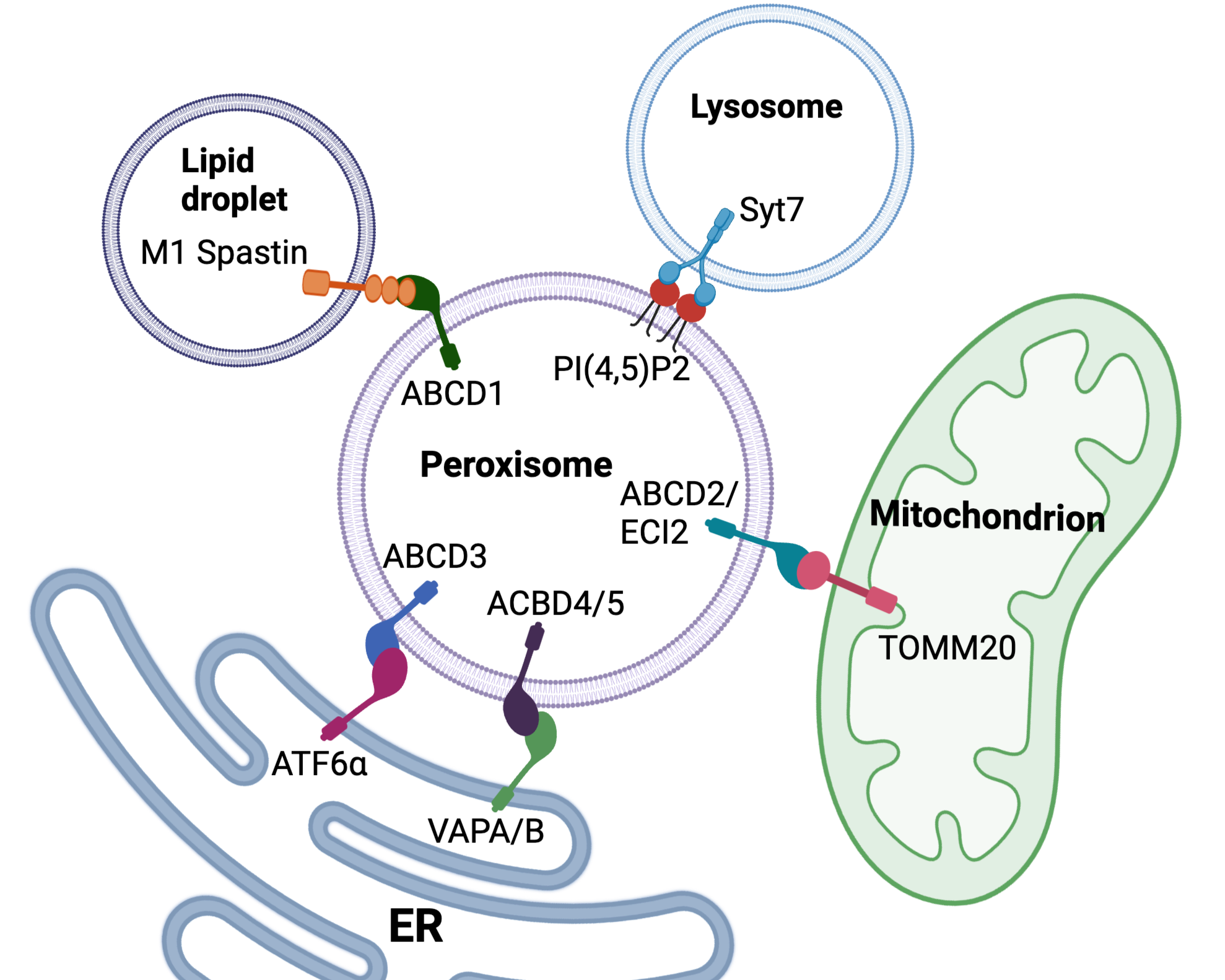
Organelle dynamics
We study peroxisome dynamics and their functional integration within the cellular network of organelles. Through live-cell imaging, proteomic profiling, and targeted molecular approaches, we investigate how these interactions are established, maintained, and remodeled in response to metabolic changes or cellular stress.
-
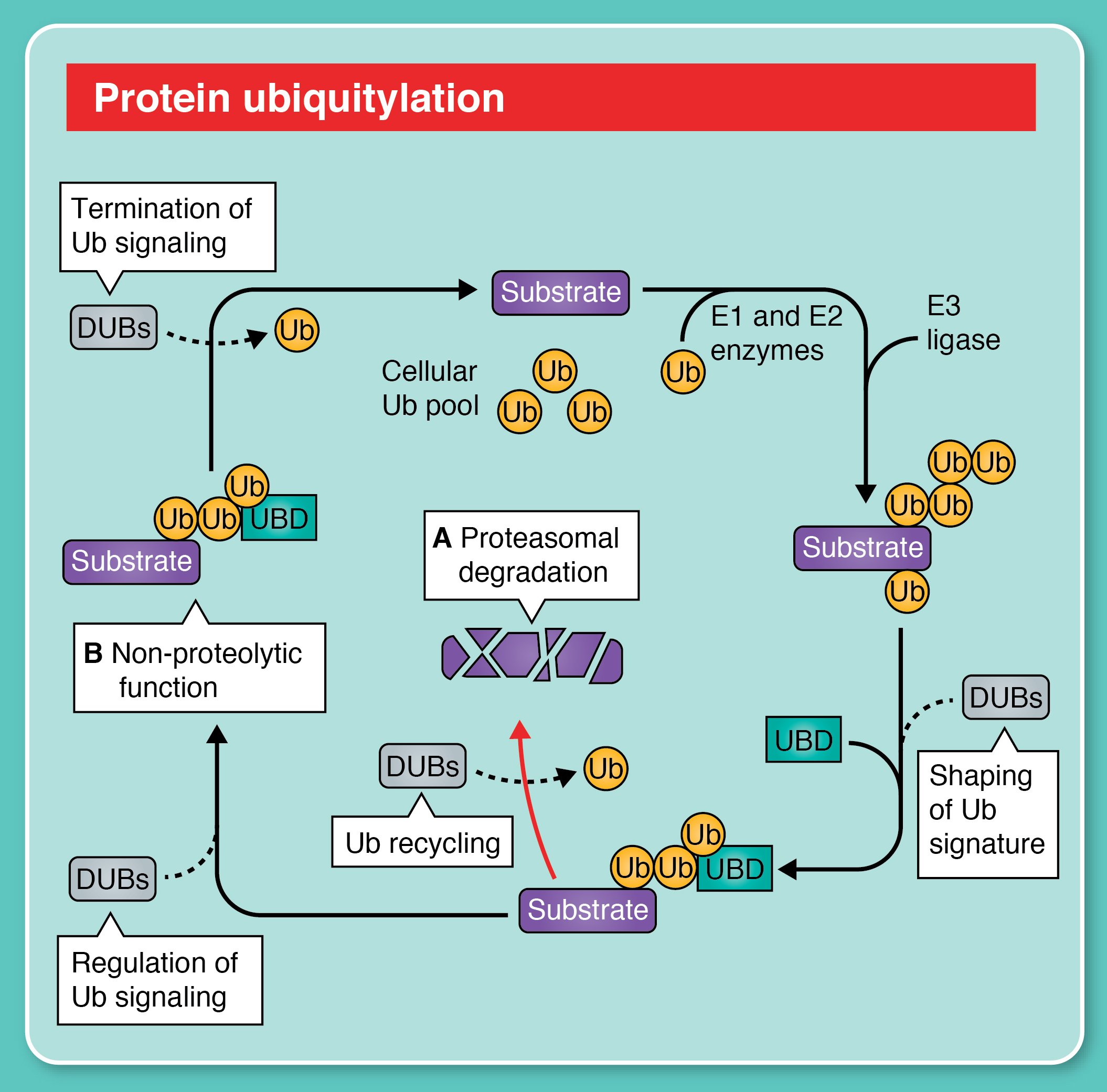
Function and regulation of DUBs
Given the complexity of ubiquitin modifications, the precise removal of ubiquitin by DUBs represents a critical challenge for the cell. DUBs maintain free ubiquitin levels, rescue proteins from degradation, and terminate ubiquitin signaling, and their activity must therefore be tightly regulated to ensure substrate and context specificity. Our lab studies the physiological roles of DUBs and how their catalytic activity is controlled in a cellular context.