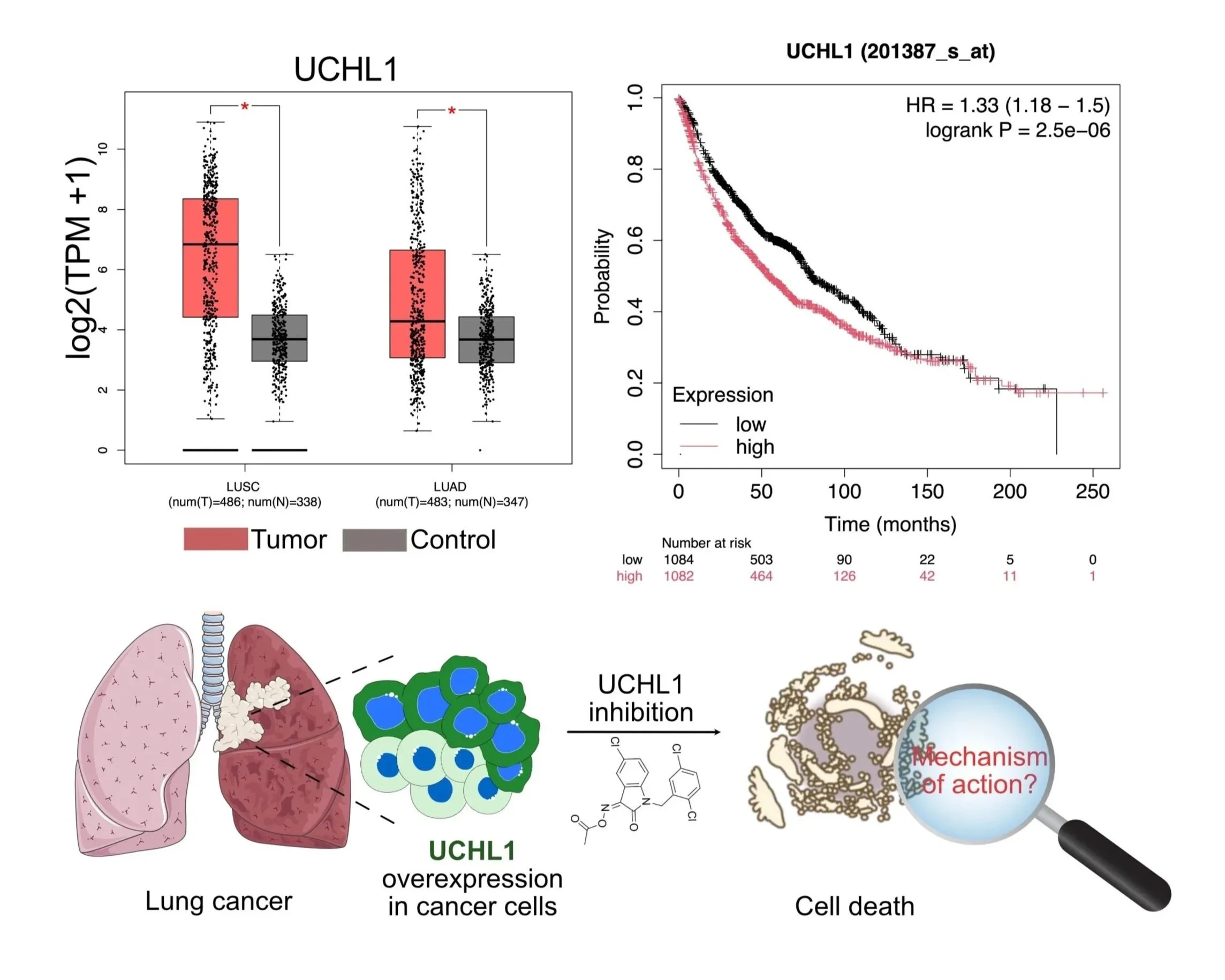
Ubiquitin-dependent regulation of V-ATPase function and mTORC1 signaling at lysosomes
Lysosomes are not only degradative organelles but also central hubs for metabolic signaling. A key outcome of this signaling is activation of mTORC1, which regulates cell growth in response to nutrients. Amino acids activate mTORC1 by recruiting it to lysosomes via Rag GTPases and the Ragulator complex, a process dependent on the vacuolar H⁺‑ATPase (V‑ATPase) . At the lysosome, V‑ATPase physically interacts with Ragulator and undergoes conformational changes in response to amino acids, acting as both scaffold and sensor for amino acid availability. Cholesterol also feeds into this pathway through NPC1 and SLC38A9, establishing the V-ATPase as an integration point for amino acid and lipid signals. We previously identified a USP32-controlled ubiquitin signal at Ragulator subunit LAMTOR1 that limits V-ATPase binding, dampens mTORC1 activation even in the presence of amino acids, and promotes autophagy (Hertel et al., Cell Rep. 2022; funded by FOR 2625 “Mechanisms of Lysosomal Homeostasis”). Together, these findings highlight ubiquitylation as an important non-proteolytic regulator of mTORC1 signaling, and our ongoing work explores how ubiquitylation further controls V-ATPase assembly and function.
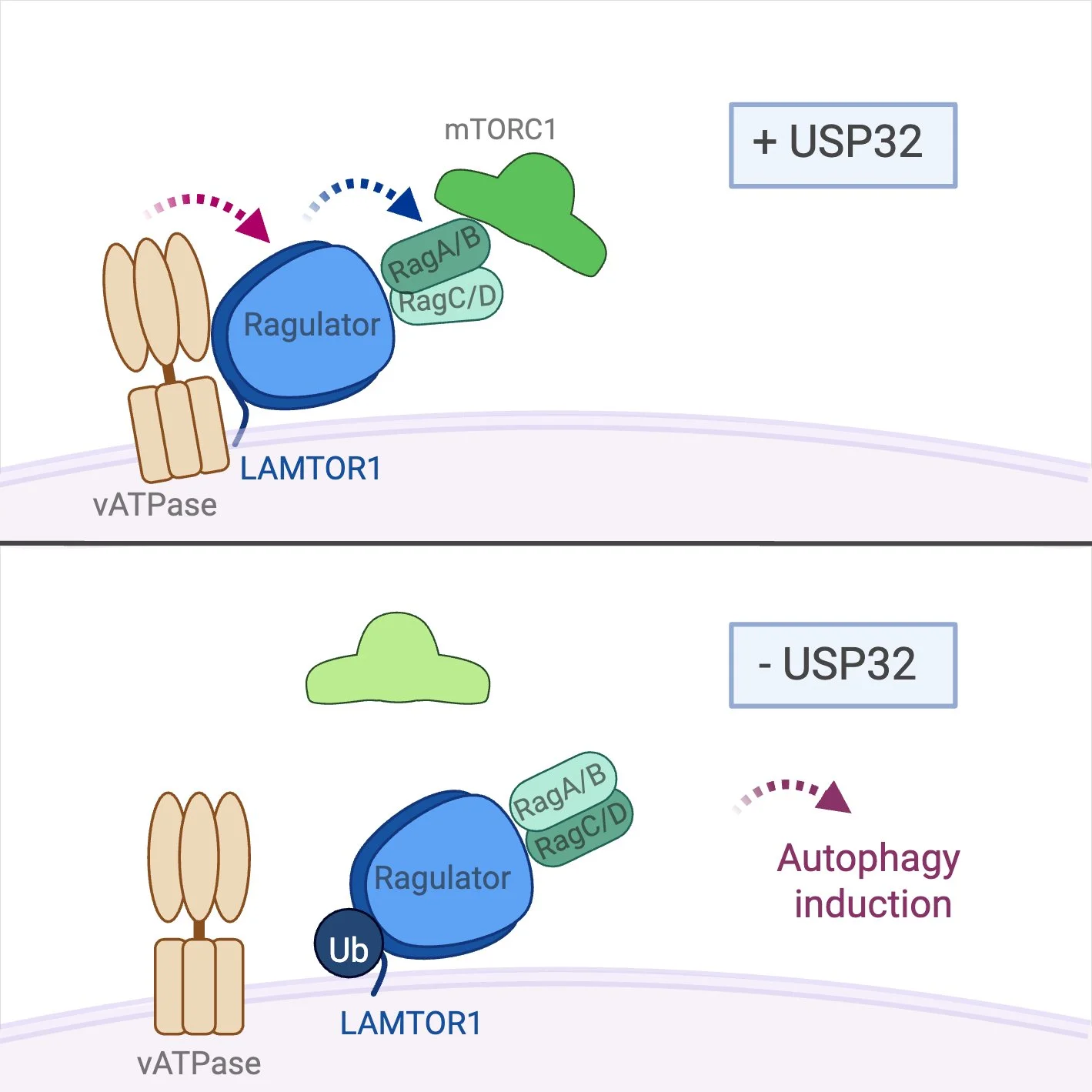
Targeting the oxidative stress response of non-small cell lung cancers
Cancer cells rely on rewired metabolism to support rapid growth, generating high levels of reactive oxygen species that necessitate strong antioxidant defenses. In many non-small cell lung cancers (NSCLC), aberrant activation of the antioxidant regulator NRF2 promotes tumor progression and therapy resistance. We proposes to target NRF2 activity indirectly by inhibiting DUBs that regulate its stability or function, offering an alternative therapeutic strategy for NSCLC.
In the framework of TRR 387 “Functionalizing the Ubiquitin System against Cancer” we investigate the role of OTUD5 and UCHL1 in regulating the antioxidative response.